Drug development: New NMI framework reduces risk of costly failure

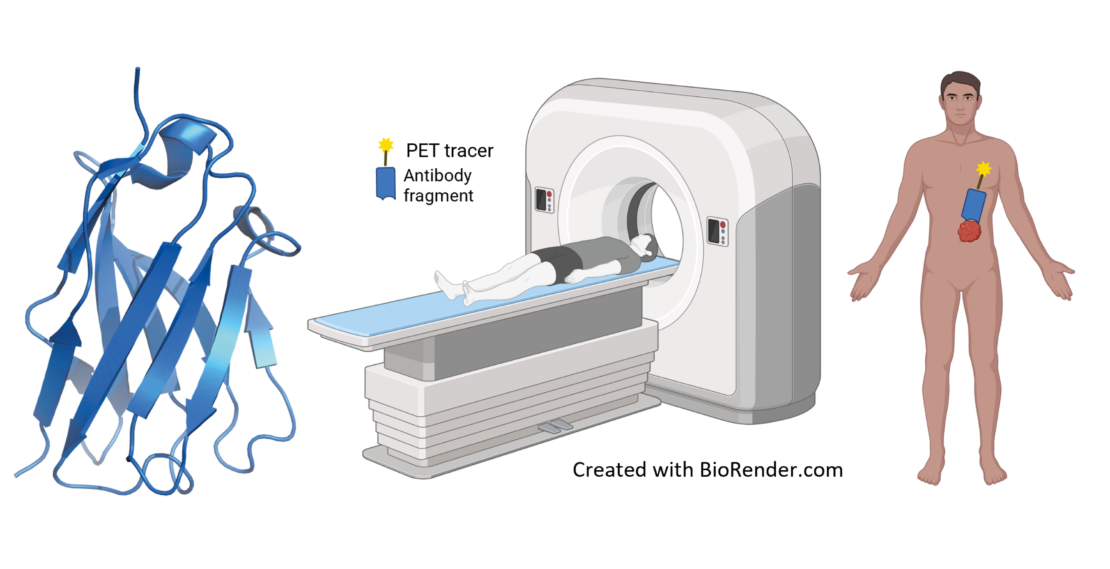
How can the risk of failure in the development of drug compounds be reduced? In a recent paper, researchers at the NMI present a newly developed framework that makes the development of drug compounds from so-called single-domain antibodies (sdAb) safer and more reliable.
Until now, drug development has generally proceeded as follows: one starts with a large number of potential drug candidates and reduces them step by step using an exclusion process—a time-consuming process that nevertheless carries the risk of late and costly failure.
An important step here is the transition from the laboratory to the clinic. The crucial question here is: Are the molecules (i.e., the sdAbs) stable enough to withstand practical application? With their newly developed framework, NMI researchers led by Anne Zeck and Philipp Kaiser can systematically identify potential weak points.
The NMI experts use various methods for their analysis. These include mass spectrometry, biophysical characterization, accelerated stability studies under defined stress conditions, and functional analysis.
The targeted application of these methods gives drug developers the opportunity to optimize their molecules at an early stage, thereby reducing the risk of late and costly failure.
To the paper: doi.org/10.3390/antib15020022