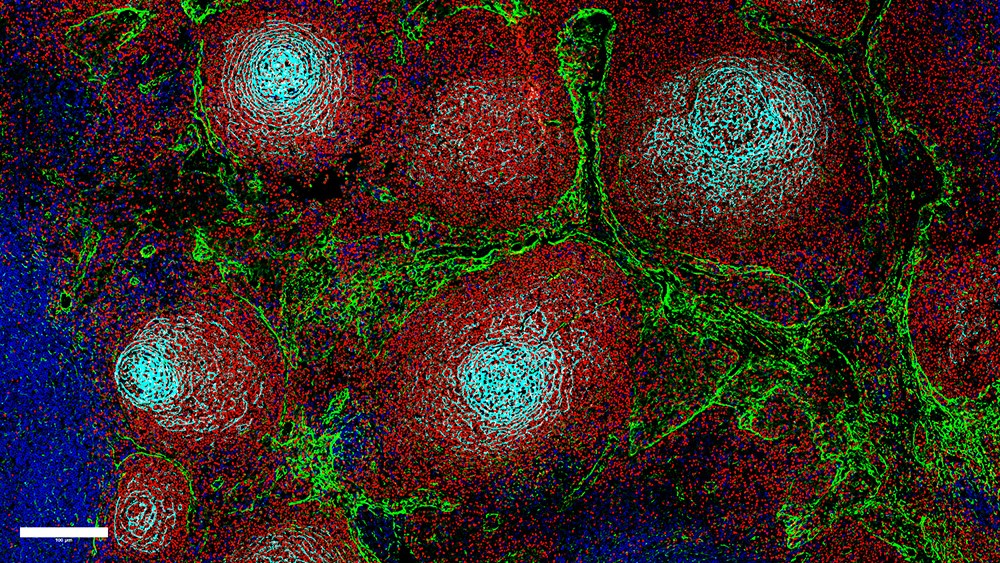
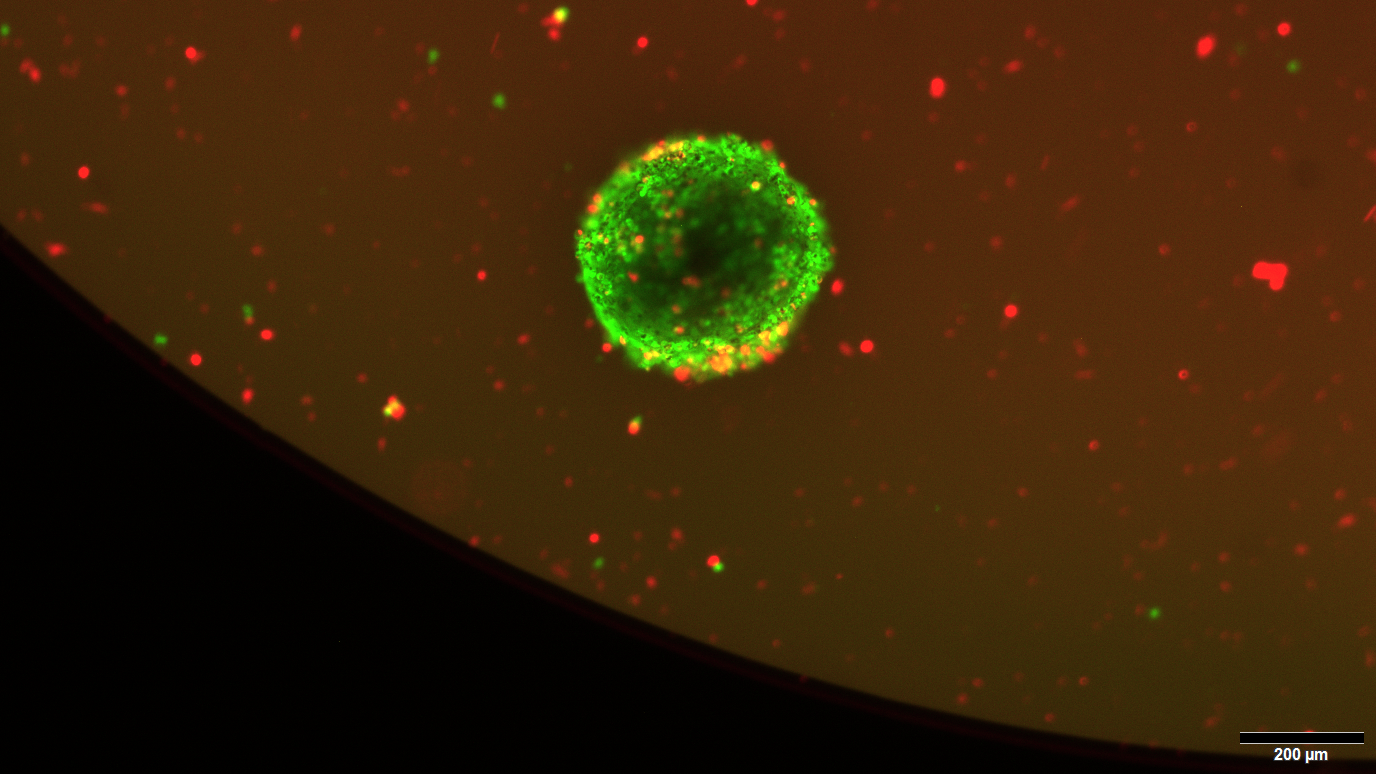
How the Epstein-Barr virus promotes its spread in the body

Many people are infected with the Epstein-Barr virus (EBV), and most are unaware of it. However, EBV can sometimes cause cancer, and this pathogen also appears to play an important role in multiple sclerosis and other autoimmune diseases. Researchers at the German Cancer Research Center (DKFZ) and the Kidney Center at Heidelberg University Hospital have discovered that EBV increases the ability of infected immune cells to migrate. In this way, the pathogen promotes its spread in the body – a discovery that may have therapeutic implications.
The Epstein-Barr virus, which belongs to the herpes virus family, was the first virus to be proven carcinogenic in humans. That was in the 1960s, and to this day it is not entirely clear how exactly the cancer-causing effect comes about. Interactions between EBV and the host organism’s immune system are likely to be decisive, as only a small percentage of those infected develop cancers such as the highly aggressive Burkitt’s lymphoma or stomach cancer.
Communication between EBV and immune cells also appears to play a role in the development of autoimmune diseases, with the result that the immune cells suddenly attack structures in the body. In MS, this attack targets the myelin sheaths of nerve cells.
In Germany, more than 95 percent of all adults over the age of 50 are infected with Epstein-Barr. Initial infections in childhood usually occur without symptoms, but infections in adulthood cause Pfeiffer’s glandular fever, a spontaneously healing inflammation of the lymph nodes and throat, in one third of cases. The virus then usually remains dormant in the body for life. However, some people develop malignant tumors or autoimmune diseases under the influence of EBV.
Viruses usually have a preference for certain cell types. The Epstein-Barr virus specializes in B cells of the immune system. B cells play a central role in both EBV-induced lymphomas and multiple sclerosis: in the former, these are the cells that multiply uncontrollably. In the latter case, B cells are significantly involved in the destruction of the nerve sheaths.
The virus goes mobile
DKFZ researcher Henri-Jacques Delecluse, who has been tracking the Epstein-Barr virus for a long time, is interested in these cases. He has now made another important discovery that could be the starting point for innovative therapies. “In cooperation with the Kidney Center at Heidelberg University Hospital, we were able to show that EBV-infected B cells exhibit characteristics of ‘homing cells,’” explains the DKFZ researcher. Homing refers to the migration of immune cells from the lymphatic vessels to specific tissues, where they are targeted at the structures of pathogens. The immunocompetent B cells then return to the lymphatic organs. “Homing cells” have the ability to overcome the barrier of the vascular endothelium – the inner wall layer of blood and lymph vessels. Normally, homing is strictly controlled and regulated by cytokines, which the cells use to communicate with each other.
Starting point for new therapies
EBV-infected B cells form a viral reservoir that remains in the human body for life. Because the viruses trigger the targeted migration of infected B cells, these spread throughout the body – and with them the viruses. The Heidelberg research group identified two proteins produced by EBV that trigger the homing behavior of B cells. “The viral proteins EBNA2 and LMP1 increase the activity of pro-inflammatory cytokines such as CCL4, which have been shown to be relevant in multiple sclerosis,” reports Henri-Jacques Delecluse. ”As a result, the infected B cells divide and swarm out. The EBV-manipulated B cells overcome the barrier of the vascular endothelium and penetrate into the brain, among other places. We have also been able to show that this process is controlled by MS-relevant messenger substances. These include the CCR1 receptor, which has been shown to play an important role in multiple sclerosis.”
The DKFZ researchers’ discovery opens up new therapeutic perspectives. “Deciphering the reaction cascade that triggers EBV-infected B cells to enter homing mode offers the possibility of targeted intervention to prevent B cell migration,” explains first author Susanne Delecluse. Using various approaches, the research team has already succeeded in blocking EBV-induced migration of B cells in animal models. “Using specific inhibitors,” says Susanne Delecluse, “we were able to reduce the survival rate of EBV-infected B cells and prevent them from spreading in the body, including to the brain.”
If this also works in humans – which has not yet been demonstrated – the approach could potentially be used to prevent autoaggressive damage to the nerve sheaths in multiple sclerosis.
Publication: S. Delecluse et al. Epstein-Barr virus induces aberrant B cell migration and diapedesis via FAK-dependent chemotaxis pathways. Nature Communications 2025, DOI: 10.1038/s41467-025-59813-z