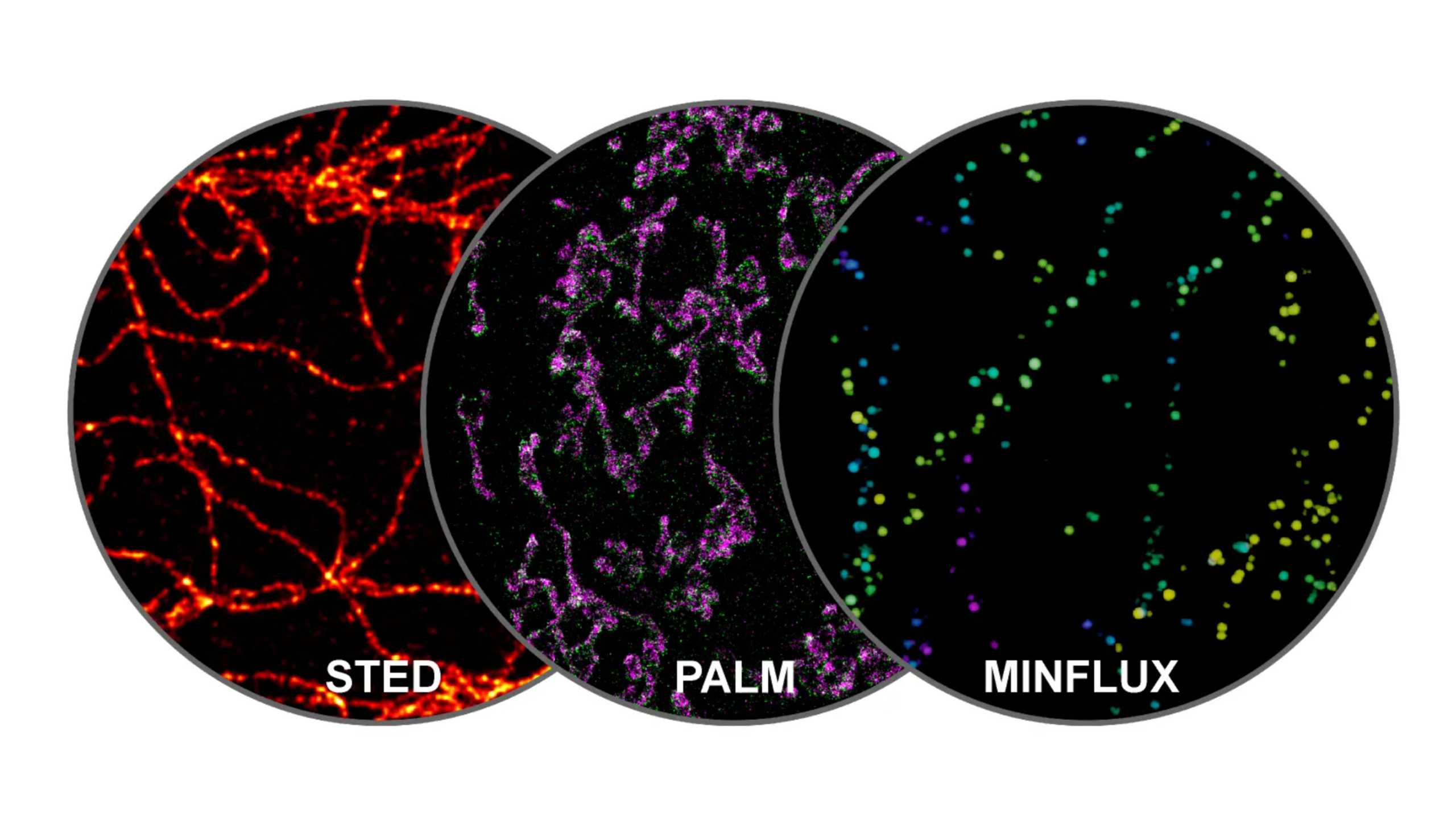
Shapes, signals, and boundaries: how cells turn physics into information

Insights from theoretical physics are helping scientists understand how living cells process information and use it to self-organise

A flock of birds, an ant colony, heavy traffic on a highway – what do they all have in common?
In addition to being things you wouldn’t want to suddenly find yourself in the middle of, they are all examples of self-organising systems – systems that function as a whole without a central controller directing motions.
Our cells and tissues are also self-organising in many ways. Membranes and filaments assemble themselves, proteins and enzymes find their way to where they are needed, and masses of undifferentiated cells resolve into perfect tissue layers inside an embryo. How cells achieve such feats of coordination while living in inherently noisy environments is little understood.
Researchers from the Erzberger Group at EMBL Heidelberg have been investigating these phenomena by applying theoretical physics approaches. They focus in particular on how cells and tissues process and use information from their surroundings – including the physical forces acting on them – to self-organise in space and time.
“Mechanisms that arise from fundamental physical principles – conservation laws, geometry, and unavoidable constraints – are often universal across scales and systems,” said Anna Erzberger, Group Leader. “As a result, theoretical approaches can uncover general features of biological organisation and make concrete, quantifiable predictions.”
How cells make binary decisions in crowded environments
One of the scientists in the Erzberger Group studying this is PhD student Jenna Elliott. Following an undergraduate degree in physics, Elliott moved into biophysics for her PhD, inspired by the versatility of biological materials. Among other things, she is fascinated by how biological systems process information gleaned from their surroundings.
Cells exist in an environment that is both noisy and complicated. There are many things they can potentially pay attention to, such as various molecules and chemical entities, physical forces like pressure and tension, and their location in space with respect to their neighbours. Given this, the scientists hypothesised that cells might apply some kind of filter when they need to make decisions, just like filters can be used in photography to remove extraneous information.
“Usually, people think of cellular information as biochemical signals that go up or down over time, like a volume knob,” said Elliott. However, sometimes the cell needs to process information more like an ‘on-off’ switch, especially when key decisions need to be made, for example, which direction to move in or when to divide.
In particular, Elliott was interested in understanding how cells compute information stored in spatial signals, for example, how close a cell is to a physical object. One way to encode such information is through the interactions between molecules distributed at the boundary with the environment, e.g. at the cell surface.
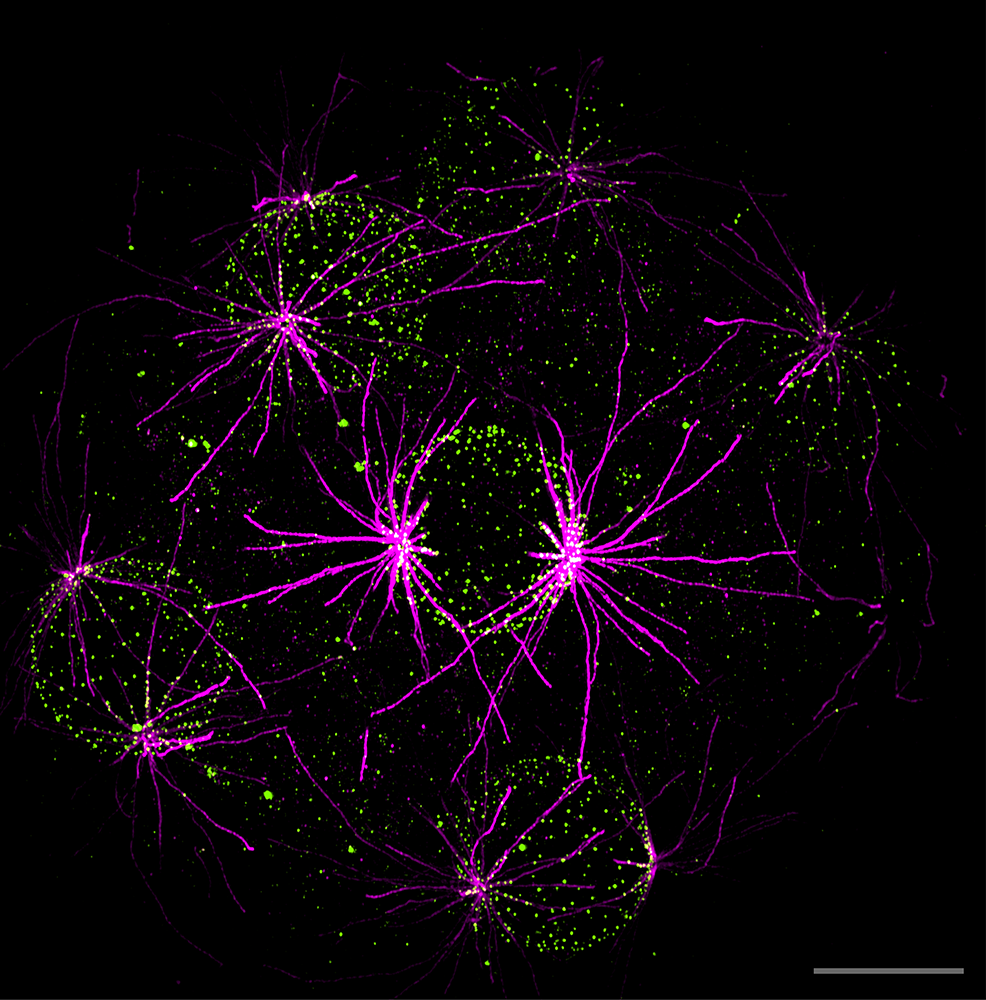
Elliott and Erzberger investigated how certain membrane-embedded particles (such as proteins) redistribute themselves on the surface in response to nearby structures they can interact with. The scientists found that when such particles repel each other within the surface, their distribution can effectively apply a ‘threshold’, converting noisy signals from their surroundings into a binary, ‘yes-no’ answer.
However, is this applicable in an actual living cell? To check this, the team turned to collaborators from Gautam Dey’s group at EMBL, who study nuclear evolution in single-celled microorganisms. Hiral Shah, an EIPOD postdoc on Dey’s team, had collected detailed images of a unicellular microorganism called S. arctica, showing the distribution of nuclear pore complexes (NPCs) on the surface of the nucleus. These complexes act as gateways between the nucleus and the rest of the cell, allowing transport of important molecules. In S. arctica, the NPCs line up along microtubule filaments that surround the nucleus, raising the possibility that they might serve as a conduit for information between the cytoskeleton and the interior of the nucleus.
“We traced the microtubules and the NPCs and found that the NPC distribution follows our theoretical predictions,” said Elliott. The researchers thus found that spatial patterns in cells can provide filtered, simplified information about surrounding structures.
“Biology has solved problems that currently plague synthetic soft matter computation,” said Elliott. “Living systems can ‘compute’ in noisy, complicated environments without electronics. If we understand these physical rules, we can design synthetic materials that adapt or compute. Soft materials are also much easier to mesh with human tissue for medical applications than silicon chips.”
For the next stage of her career, Elliott is planning to continue exploring the border between biology and materials science to see whether physical systems can replicate the solutions that biological systems have evolved over millennia.
Feedback loops allow shape and signalling to interact
Unlike Elliott, Tim Dullweber, a former PhD student in the Erzberger Group and currently a postdoc at the University of Basel, started his scientific journey with wet-lab research, through double undergraduate degrees in molecular medicine and physics. However, he had always been fascinated by theory, and decided it was “now or never” when it was time to choose a direction for his doctoral studies.
Dullweber was also intrigued by the physics at the boundaries of biological systems, such as cell membranes. As the main focus for his PhD, he wished to understand the dynamics that arise when one couples the mechanical properties of cells with biochemical signalling pathways at such boundaries.
Cells often use a series of biochemical reactions between specialised molecules to transmit and amplify information. A well-known example is the Notch signalling pathway, which plays an important role in embryonic development and tissue repair, and is dysregulated in many cancers.
Unlike most other signalling pathways, the Notch signalling pathway involves a ligand and a receptor that are both embedded in the cell membrane. These molecules therefore only interact across cell–cell contacts, i.e. in regions where the membranes of neighbouring cells are held close together by the cells’ mechanical machinery. When and with whom a cell communicates is therefore determined by the geometry and mechanics of the cell surface. In turn, signalling interactions can trigger changes in cell surface mechanics, leading to feedback effects.
To theoretically investigate the collective dynamics arising from this coupling between biochemical signals and mechanics, Dullweber and his colleagues decided to compare the cells’ behaviour to that of droplets or foams, whose shape is determined by their surface tension.
In contrast to passive materials, cells can respond quite quickly to signals and actively change their surface properties. By coming up with a mathematical description for such adaptive droplets, the researchers discovered that such materials can oscillate spontaneously, as well as undergo rapid switching between distinct shapes and signalling levels.
For example, when signalling increases the size of a cell–cell contact, positive feedback arises. This can be used by the cell to suppress weak signals and amplify strong signals. “A small contact area means fewer molecules can interact; a large contact area means more signalling,” explained Dullweber.
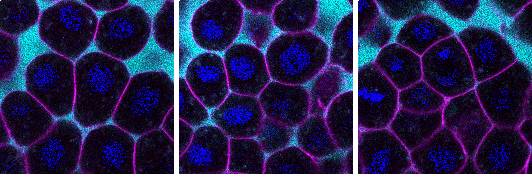
Positive feedback can also lead to sharp transitions between distinct cellular states. A biological system in which sharp, stable boundaries between tissue regions are essential is the developing embryo. Together with collaborators Camilla Autorino and Nicoletta Petridou, the team then tested their theoretical predictions by measuring cell–cell contacts and signalling states in the zebrafish embryo. They found that the shape transitions their model predicted can help establish the boundary between the internal and external tissues of the embryo during development.
Both these studies reveal how biological materials, like cells and tissues, use physical information to control and coordinate fundamental biological processes. In addition to helping us understand how cells make sense of their environments, these studies could have applications in the field of bio-inspired engineering, for example, by providing design principles for smart materials that respond to changing environments.
“I think while it’s clear that physics can help us understand biology, we also see in these studies how biology can inspire new physics,” said Dullweber.
Theory@EMBL
Introduced in EMBL’s 2022–26 Programme, the Theory@EMBL research programme promotes theory-guided paths to discovering, understanding, and conceptualising the underlying principles of complex and dynamic biological systems at all scales, from molecules to organisms to ecosystems. Several EMBL groups, including the Erzberger, Graf, and Torres-Sánchez groups are working on developing novel theoretical frameworks that build new understanding of biological systems and processes and solve challenging biological problems. Learn more here.
Source article(s)
Repulsive Particle Interactions Enable Selective Information Processing at Cellular Interfaces
Elliott J., et al.
Physical Review Letters 21 November 2025
10.1103/ywr6-mzz5
Shape Switching and Tunable Oscillations of Adaptive Droplets.
Dullweber T., et al.
Physical Review Letters 19 September 2025
10.1103/1cq4-x499
Feedback between microscopic activity and macroscopic dynamics drives excitability and oscillations in mechanochemical matter.
Dullweber T., et al.
Physical Review E 19 September 2025
10.1103/mmz3-kbrv