Study shows how lymph node architecture affects cancer growth
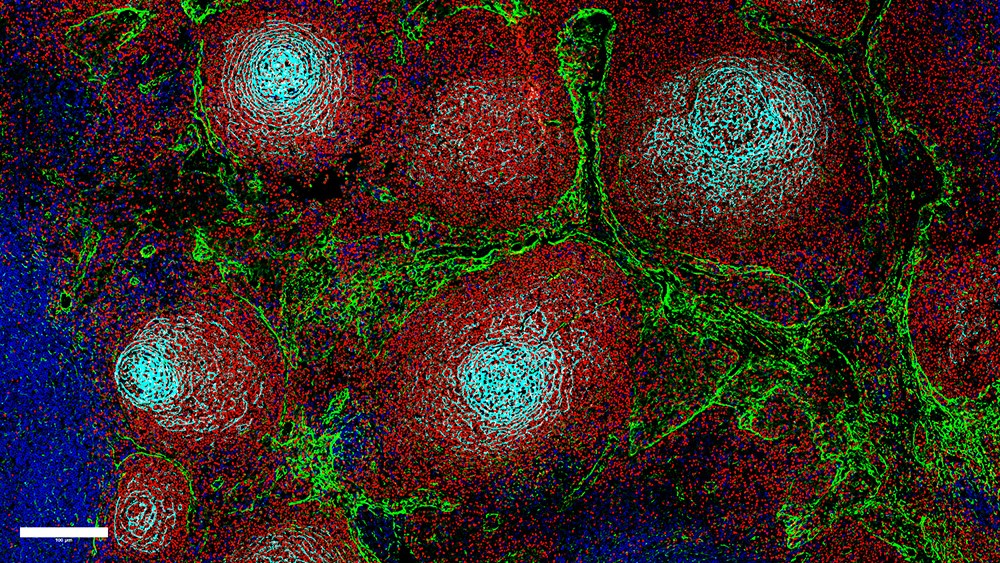
A new study maps the organisation of immune and stromal cells in lymph nodes and shows how loss of this organisation affects prognosis in certain lymphomas

Summary
- Lymphomas are a group of heterogeneous cancers of the lymphatic system and are increasing in incidence across the globe.
- A new study maps for the first time how immune cells are organised in lymph nodes and explores how this architecture is affected in different types of lymphomas.
- The researchers found that an ‘inflammatory vicious cycle’ drives this loss of organisation, where cascades of pro-inflammatory signals lead to a breakdown in lymph node tissue structure.
- The findings have implications for long-term prognosis in cancer patients and can potentially open up new therapeutic avenues.
Researchers from EMBL Heidelberg, Heinrich Heine University Düsseldorf (HHU), and Düsseldorf University Hospital (UKD) have succeeded for the first time in mapping the organisation of immune and stromal cells in human lymph nodes. With this, they were able to show how the architecture of the lymph node gradually breaks down in cancers like malignant lymphomas, leading to worse prognosis. The results were published in the journal Nature Cancer.
The study was conducted in collaboration with the German Cancer Research Center (DKFZ), the Max Delbrück Center for Molecular Medicine in Berlin, and the University of Basel, among others.
Exploring tissue architecture in lymph nodes
Lymph nodes are central hubs of the immune system and play an important role in defending against infections and tumours. For this, immune cells (B cells and T cells) must be spatially organised in the lymph node tissue, for example, in areas called B-cell follicles and T-cell zones. This is controlled by support cells called stromal cells. Stromal cells release messenger substances, known as chemokines, which provide guiding signals that immune cells use to find their designated places in the lymph node.
In B-cell lymphomas, the internal order of the lymph node tissue is disrupted in different ways depending on the type of lymphoma. While the basic tissue structure remains intact in slow-growing lymphomas, such as follicular lymphoma (FL), the spatial order collapses completely in aggressive lymphomas, such as diffuse large B-cell lymphoma (DLBCL). Until now, it was largely unclear why these typical growth patterns develop.
The new study, for the first time, systematically maps these processes in human lymph nodes. Using single-cell analyses and spatial tissue mapping, the scientists were able to trace the factors that cause the lymph node architecture to increasingly disintegrate in lymphomas.
“This project was really shaped by collaboration – four groups and three first authors all bringing different perspectives and skills, from clinical insight to state-of-the-art technologies and computational analysis,” said Anna Mathioudaki, former PhD student in the Zaugg Group at EMBL Heidelberg and currently a postdoc at DKFZ. “It was also just a genuinely fun team to work with. I learned a huge amount along the way, and many of the ideas that came out of this study have gone on to shape the bigger scientific questions I continue to pursue.”
Inflammatory vicious cycle in lymphomas
The data show that stromal cells are the ‘architects’ of the lymph node. The researchers demonstrated that central chemokine signalling in specialised stromal cells undergoes fundamental changes in the context of lymphoma, causing the spatial organisation of the lymph node to gradually collapse. These changes in stromal cells are reflected in the growth pattern of lymphomas. In the slow-growing FL, the proportions of B-cell follicles and T-cell zones shift, and the areas remain largely spatially separated. In the more aggressive DLBCL, important control signals and thus the tissue structure are largely lost.
The study identifies an inflammatory vicious circle as the driving mechanism: as part of the immune response in the tumour environment, T cells produce pro-inflammatory messenger substances, known as interferons, which cause stromal cells to change their chemokine production. Instead of structure-forming signals, inflammatory chemokines dominate, which, in turn, attract further inflammatory cells. The loss of lymph node organisation in lymphomas is therefore not a passive effect of tumour growth, but is actively driven by inflammatory processes in the tumour environment.
For patients, this reprogramming of stromal cells leads to poorer survival chances. The study was able to show in large cohorts that a loss of structure-giving chemokines is associated with an unfavourable prognosis.
“Our work links tissue organisation directly to disease outcome, which shifts the perspective from focusing only on cancer cells to also considering the tumour microenvironment, including structural cells,” said Judith Zaugg, Group Leader at EMBL Heidelberg and a senior author of the study. “For cancer research more broadly, it highlights how important it is to study the role of stromal cells on reshaping tissues in ways that influence immune responses and therapy outcomes. The findings also suggest new avenues for biomarkers of tissue organisation rather than tumour cells alone.”
In addition, the findings also open up new potential therapeutic approaches. “The study results show us that stabilising stromal cells or specifically modulating inflammatory signals could be a promising new therapeutic approach,” said Sascha Dietrich, Director of the Department of Hematology, Oncology and Clinical Immunology at UKD, former MMPU group leader, and a senior author of the study.
The above text was translated and adapted from a press release from the Heinrich Heine University, Düsseldorf. Read the original text here.
The study was conducted as part of a national and international collaboration. In addition to the people named above, the following individuals played a leading role: Felix Czernilofsky (Heidelberg University Hospital), Lea Jopp-Saile, Simon Haas (both Max Delbrück Center for Molecular Medicine, Berlin), Daniel Hübschmann (DKFZ). Christina Schniederjohann, Nora Liebers, Peter-Martin Bruch, Marc Seifert (all from the Department of Hematology, Oncology, and Clinical Immunology) and Jörg Distler (Department of Rheumatology) were also involved at the Düsseldorf site.
Source article(s)
Reprogramming of stroma-derived chemokine networks drives the loss of tissue organization in nodal B cell lymphoma.
Czernilofsky F., Mathioudaki A., Jopp-Saile L. et al.
Nature Cancer 25 March 2026
10.1038/s43018-026-01136-z