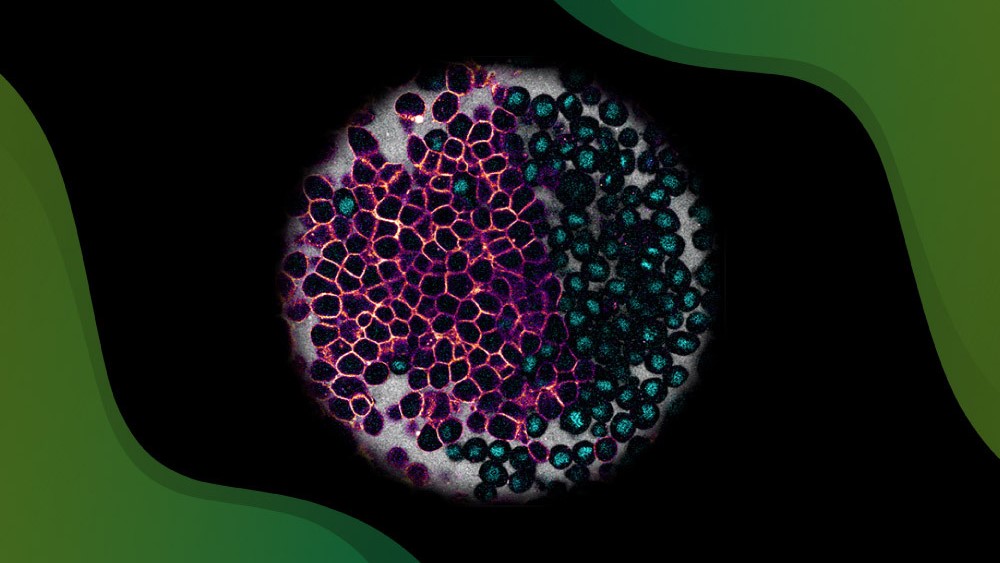
With lipid nanoparticles (LNPs), PROGEN offers an alternative drug delivery tool for the gene therapy market

Non-viral platform for efficient drug transfer to facilitate clinical developments
Heidelberg, 15.01.2025 – PROGEN, leading manufacturer of Adeno-Associated-Virus (AAV) analytical tools for gene therapy research and development, announces the launch of LNPs for the versatile delivery of therapeutic agents into cells.
In the past decade, PROGEN has driven progress in gene therapy by providing reliable ELISA assays for AAV vector characterization and titer determination that enable safe and reliable gene transfer. Answering the market need for a non-viral drug delivery system that overcomes common hurdles, such as nucleic acid degradation, insolubility or inefficient administration, the company is now offering customized LNP kits in addition to its comprehensive analytical AAV product range.
LNPs are composed of a blend of lipids, formulated to transport nucleic acids, proteins or other small molecules efficiently into target cells. Their versatile structure provides robust protection from degradation, ensures high cell internalization, and a rapid delivery rate of encapsulated therapeutic candidates (e.g. APIs, anionic peptides, proteins, or mRNA). The efficiency of the biocompatible, loaded nanoparticles can be monitored with real-time fluorescent imaging. By improving solubility, pharmacokinetics, pharmacodynamics and overall therapeutic activity, LNPs have been designed to accelerate clinical development and PROGEN can collaborate with CDMOs to provide GMP-manufactured formulations as a scalable platform for clinical research organizations worldwide.
“The introduction of LNPs is a significant step forward in our commitment to supporting scientists in advancing gene therapy”, says Katja Betts, CEO. “We are excited to be able to offer a non-viral kit platform for the safe and efficient delivery of various cargoes, in addition to our AAV analytical tools‘‘, adds Maik Lander, CEO.
PROGEN intends to continue driving progress in the gene therapy field with a global market value of 17.4B USD in 2023 (ExpertMarketResearch) and a projected 18% share of LNPs in 2036 (insightaceanalytics).
About PROGEN
PROGEN is dedicated to supporting scientific discoveries and advancing research in gene therapy. Our mission is to help accelerate the development of safe, effective therapies to improve the lives of those affected by disease. With a team of experts and global partnerships, we go beyond manufacturing antibodies, proteins, and ELISA kits. We focus on understanding the needs of scientists to create high-quality, tailored solutions that address research challenges across academia, biotech, and pharma. At PROGEN, we strive to be a trusted partner, driving innovation and progress in groundbreaking research in gene therapy.