Spliceosome: How Cells Avoid Errors When Manufacturing mRNA

Structural biologists provide a first-time look at the atomic level into the quality control mechanism of this complex molecular machine
A complex molecular machine, the spliceosome, ensures that the genetic information from the genome, after being transcribed into mRNA precursors, is correctly assembled into mature mRNA. Splicing is a basic requirement for producing proteins that fulfill an organism’s vital functions. Faulty functioning of a spliceosome can lead to a variety of serious diseases. Researchers at the Heidelberg University Biochemistry Center (BZH) have succeeded for the first time in depicting a faultily “blocked” spliceosome at high resolution and reconstructing how it is recognized and eliminated in the cell. The research was conducted in collaboration with colleagues from the Australian National University.
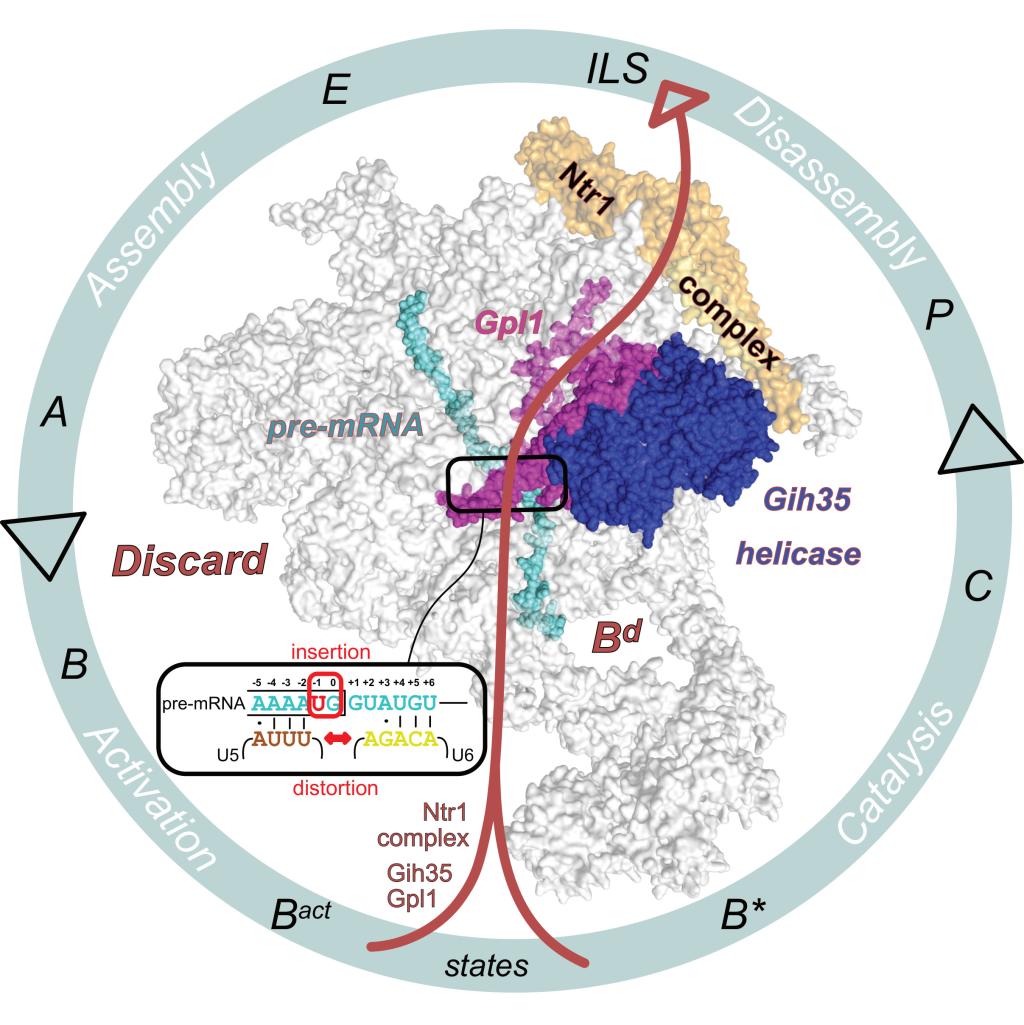
The genetic information of all living organisms is contained in the DNA, with the majority of genes in higher organisms being structured in a mosaic-like manner. So the cells are able to “read” the instructions for building proteins stored in these genetic mosaic particles, they are first copied into precursors of mRNA, or messenger RNA. The spliceosome then converts them into mature, functional mRNA. To do this, this large protein-RNA complex, which is located in the cell nucleus, removes non-coding sections (introns) from mRNA precursors and links the coding sections (exons) to form a continuous strand of information. Errors in this process, also known as splicing, are one of the main causes of inheritable genetic disorders and are associated with neurodevelopmental disorders and diseases such as cancer. It was known that the spliceosome has quality control mechanisms, but the mechanistic details were not understood.
For their experiments, the Heidelberg researchers led by BZH director Prof. Dr Irmgard Sinning used the fission yeast Schizosaccharomyces pombe, a model organism frequently used in cell biology. Using molecular markers, defective spliceosomes were identified, purified, and structurally examined via cryo-electron microscopy. “The largely stable structure of the spliceosome center enabled us to obtain high-resolution information. This means that a spliceosome discarded during cellular quality control can be represented at the atomic level for the first time,” states the structural biologist. “However, analyzing the components flexibly bound to the periphery of the spliceosome was a major challenge for our work,” explains Dr Komal Soni from the BZH.
Based on this structural information, the scientists were able to understand which errors occur during splicing, how the spliceosome recognizes faulty processes and subsequently aborts the splicing, thereby sorting out the faulty complex. Using the detailed structures, the researchers were also able to model the underlying molecular mechanisms. The proteins involved in this process of cellular quality control are conserved in eukaryotic organisms from fission yeast to humans. The scientists therefore assume that the mechanisms for recognizing and sorting out faulty spliceosomes have remained largely unchanged over the course of evolution.
The research was carried out as part of a long-term collaboration between the teams of Prof. Sinning and Prof. Dr Tamas Fischer, who specializes in RNA surveillance at the Australian National University in Canberra. Prof. Dr Henning Urlaub’s research group at the Max Planck Institute for Multidisciplinary Sciences in Göttingen also participated. The work was funded by the German Research Foundation and the Australian Research Council. The results of the research were published in “Nature Structural & Molecular Biology”.
Original publication
K. Soni, A. Horvath, O. Dybkov, M. Schwan, S. Trakansuebkul, D. Flemming, K. Wild, H. Urlaub, T. Fischer & I. Sinning: Structures of aberrant spliceosome intermediates on their way to disassembly. Nature Structural & Molecular Biology (20 January 2025),




